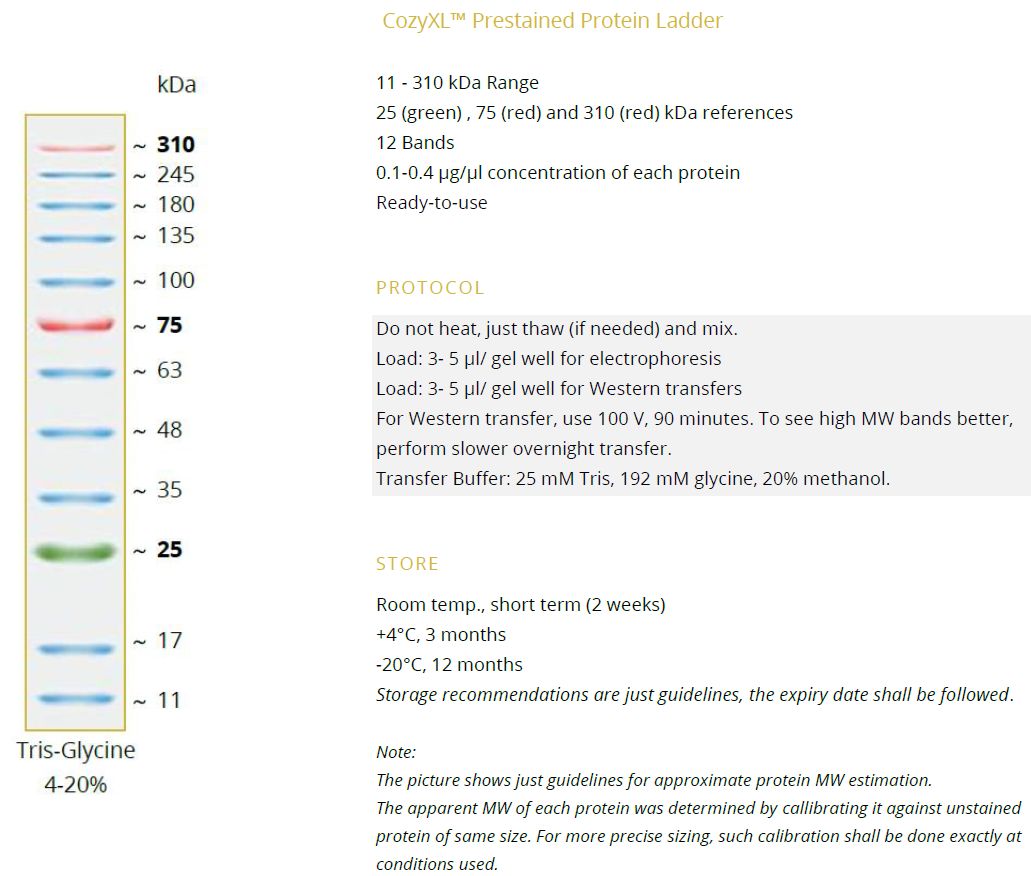
NEW StainIN™ eco-RED Nucleic Acid Stain - a less toxic alternative to ethidium bromide
Applications
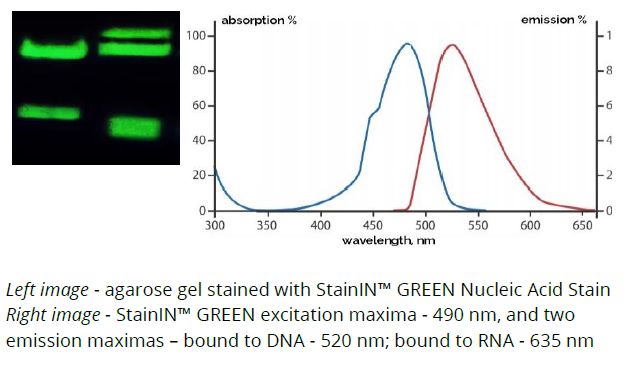
- Staining of DNA or RNA in agarose and polyacrylamide gels
- ssDNA, dsDNA and RNA visualization under the UV light
- ssDNA, dsDNA and RNA visualization under LED light
Benefits
- Replaces discontinued StainIN™ RED Nucleic Acid Stain NAS0101
- Safe, DMSO-free, economical alternative to ethidium bromide
- Universal – in gel staining, or post- run staining, no de-staining.
- Ambient temperature storage (protected from light)
The carcinogenicity and a high toxicity of a widely used gel stain Ethidium Bromide (EtBr) is one of the biggest safety and environmental problems in the labs where gel electrophoresis is a daily routine. Therefore, there is a need for a safer ethidium bromide alternatives to be used in laboratories. The novel less toxic nucleic acid dyes used for agarose gel electrophoresis address this ethidium bromide problem and minimize environmental concerns.
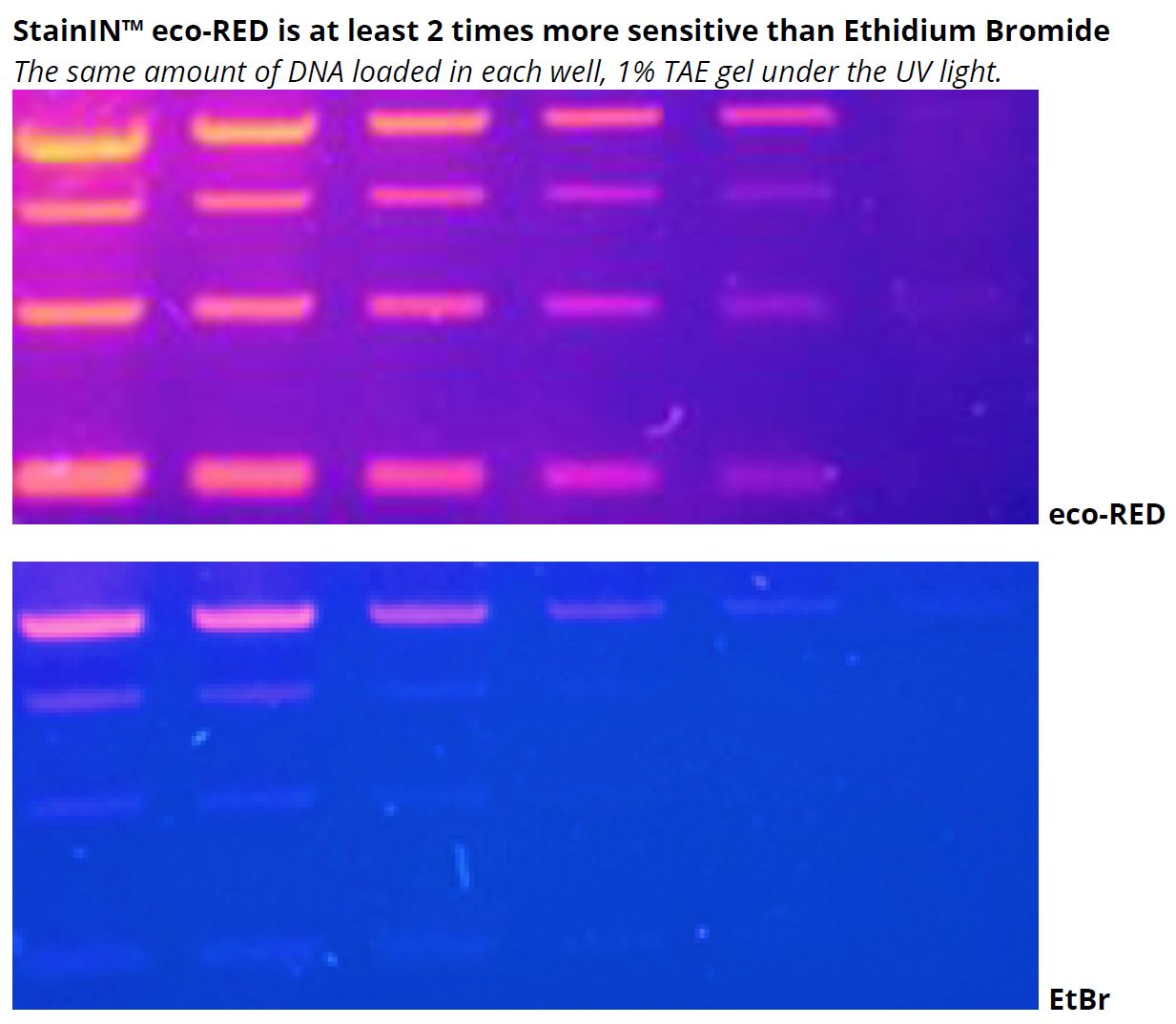
highQu StainIN™ eco-RED Nucleic Acid Stain provides a significantly safer alternative to ethidium bromide while delivering up to several times higher detection sensitivity. Same like ethidium bromide, the stain is used during the process of electrophoresis in an agarose gel or PAAG and (optionally) in the electrophoresis running buffer, or alternatively, for a post-run staining of the gels in a tray with the buffer.
Despite having a proven lower mutagenicity, all nucleic acid stains shall be handled with care, as they interact with DNA and RNA. Used electrophoresis buffer with a stain as well as melted gels shall be run through approved (carbon) filters. If the absence of residual fluorescence is confirmed, the liquids can be disposed with plenty of water down the drain.
Alternatively, all used liquids can be autoclaved or heated as the StainIN™ eco-RED Nucleic Acid Stain degrades during the long exposure to above 100ºC temperature.
The safety office shall be consulted periodically to match local regulations, as these regulations vary and change. Despite of tests described in this report, the seller of the listed stains and other reagents does not take any responsibility for the possible damage resulting from handling any sold chemical or reagent.
Frequently asked questions about the new Nucleic Acids Stain
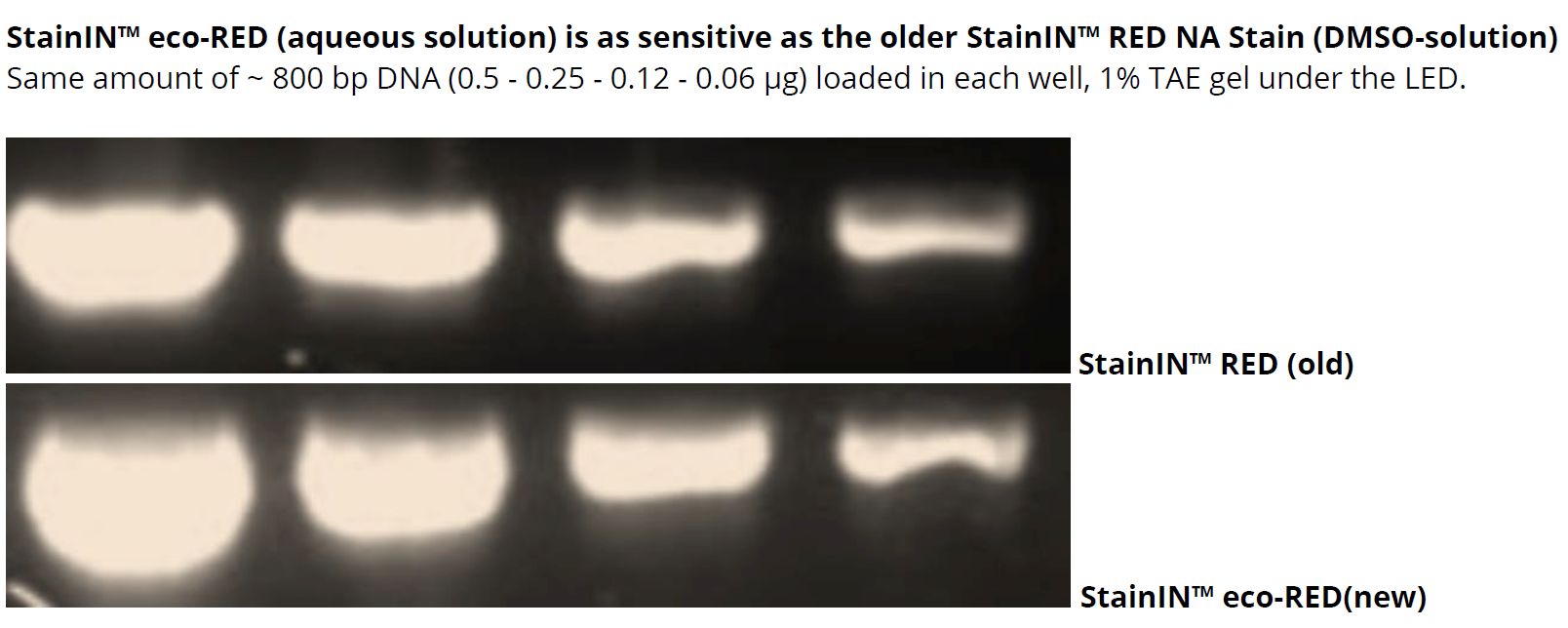
1. What are the advantages of the new StainIN™ eco-RED NA Stain over the old version StainIN™ RED?
- The new stain is purely aqueous solution. The old StainIN™ RED had DMSO as solvent.
- The new stain is room temperature stable, shipped at ambient temperature. Store it at your electrophoresis bench. The old StainIN™ RED was stored at +4°C.
- StainIN™ eco-RED NA Stain is more flexible – detectable under both UV and LED light.
- More convenient protocol – the stain can be used both during the gel run and as a post-run staining.
- Same or better sensitivity compared to the old stain.
2. Is the color of the solution the same as before?
The new stain is a darker shade of red.
3. What is the concentration of the StainIN™ eco-RED Nucleic Acids Stain?
It is 10,000X concentrated, however the number of applications for post run staining is difficult to calculate because multiple gels can be stained in the same staining solution.
4. What is the solvent used in the StainIN™ eco-RED Nucleic Acids Stain?
The solvent is 100% water.
5. Which stains are similar to StainIN™ eco-RED Nucleic Acids Stain?
The closest similar stains are Ethidium Bromide and GelRed (Biotium).
6. What is the sensitivity of the StainIN™ eco-RED Nucleic Acids Stain in ng/band?
The sensitivity ranges from 0.1 to 0.3 ng DNA/band or even higher, depending on factors such as the staining method used, imaging capability, detection, and gel thickness.
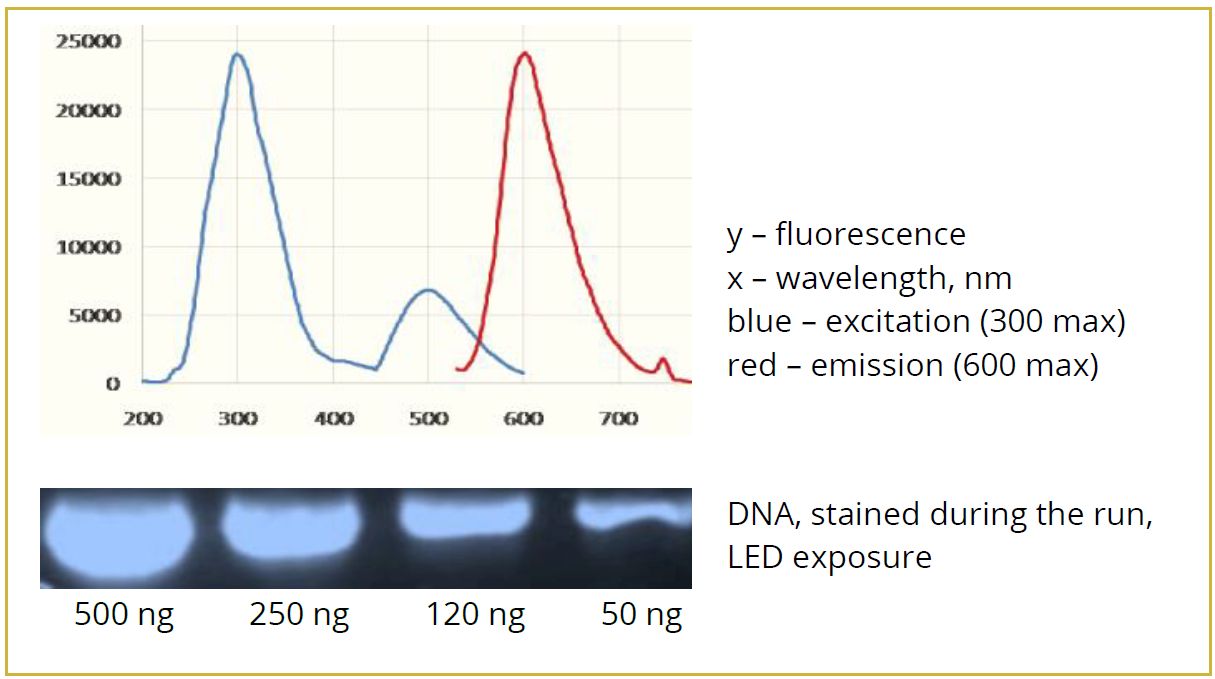
7. What is the emission wavelength of the StainIN™ eco-RED Nucleic Acids Stain?
The emission wavelength is 600 nm.
8. What is the excitation wavelength of the StainIN™ eco-RED Nucleic Acids Stain?
The excitation wavelength is 300 nm.
9. Can StainIN™ eco-RED Nucleic Acids Stain be used for in-gel and in-buffer staining?
Yes, it can be used for both in-gel and in-buffer staining post electrophoresis run.
10. How should the stain be used during a gel run in agarose gels?
Use 8-10 µl of the stain in 100 ml agarose gel solution cooled down for pouring, nothing to be added in to the electrophoresis buffer.
11. How to use StainIN™ eco-RED Nucleic Acids Stain with PAAGE gels? Same like with agarose gel, or using the post-run staining protocol.
12. Can the StainIN™ eco-RED Nucleic Acids Stain be detected under UV light?
Yes, it can be detected under UV light, with ethidium bromide or GeRed filters.
13. What color does the stained DNA appear as under UV light?
DNA appears as red-orange under the UV light.
14. What color does RNA appear as under UV light?
RNA appears as red-orange under UV light.
15. Does StainIN™ eco-RED Nucleic Acids Stain color both single-stranded and double-stranded DNA?
Yes, it can stain both single-stranded and double-stranded DNA, and RNA.
16. Can the stained DNA be detected with a blue LED?
Yes, the StainIN™ eco-RED Nucleic Acids Stain -stained DNA can be detected with a blue LED.
17. Is StainIN™ eco-RED Nucleic Acids Stain compatible with cloning?
Yes, as it can be used with LED without damaging DNA.
18. Has StainIN™ eco-RED Nucleic Acids Stain undergone toxicity testing? Yes, it is less toxic than Ethidium Bromide See the toxicity testing report.
19. Can StainIN™ eco-RED Nucleic Acids Stain be used for post-run staining?
Yes, it can be used for post-run staining.
20. Can the staining buffer (electrophoresis buffer with the NA stain) be reused?
Yes, the staining buffer can be reused several times, depending on gel thickness and the amount of DNA stained. It should be stored in the dark in a glass or plastic container. To avoid StainIN™ eco-RED Nucleic Acids Stain absorbance on plastic or glass over the prolonged storage of more than 24 hours, make a fresh staining solution every day.
21. Is the StainIN™ eco-RED Nucleic Acids Stain sensitive to light?
Yes, same as all other fluorescent nucleic acid’s stains, the StainIN™ eco-RED Nucleic Acids Stain is light-sensitive and should be protected from light.
22. Can StainIN™ eco-RED Nucleic Acids Stain be stored at +4°C?
The recommended storage conditions for this stain is ambient temperature at about +20°C.
However, due to common cooled shipments, it can be exposed for a short time to +4°C. If accidentally shipped with cooling packs at +4°C, simply mix the product to ensure the stain is completely dissolved. And keep it further only at ambient temperature.
23. What happens if the StainIN™ eco-RED Nucleic Acids Stain was put or exposed to -20°C and was frozen?
If frozen, the StainIN™ eco-RED Nucleic Acids Stain should be disposed and the new product shall be ordered. Do not use it if once frozen.
- Download Protocol and Specifications - Product Insert StainIN™ eco-RED Nucleic Acid Stain
- Ask for a Sample today
- Have technical questions? Contact us
- Download a list of Publications mentioning highQu products
- Or see how others use our products at bioz.com or google scholar
- Download Protocol and Specifications - Product Insert StainIN™ eco-RED Nucleic Acid Stain
- Download MSDS StainIN™ eco-RED Nucleic Acid Stain
- Need a lot-specific Certificate of Analysis? E-mail us at info@highQu.com
- Want custom formulations or bulk sizes? E-mail us at info@highQu.com, and check our OEM offers
- Have more specific questions? Contact us
- Download Protocol and Specifications - Product Insert StainIN™ eco-RED Nucleic Acid Stain
- Download MSDS StainIN™ eco-RED Nucleic Acid Stain
- Download highQu Catalogue of Premium Research Tools
- Download a list of Publications mentioning highQu products
- Or see how others use our products at bioz.com or google scholar
- Download Product Pricelist
- Download all highQu Product Inserts and MSDS sheets
- Have questions? Contact us